Chen Lab
Read BioMy laboratory studies neuroinflammation. In particular, we investigate the roles of molecular pathways and innate immune processes related to microglia in multiple sclerosis but also other diseases where CNS inflammation is prominent. A key aspect of our research is in deciphering complex in vivo biology by using innovative molecular imaging technologies, targeted molecular treatments, advanced experimental models as well as human trials. Ultimately we hope advance the diagnosis and treatment of these diseases by developing novel imaging and treatment strategies.
Recent Publications
Abou Haidar E, Prabhakar S, Geffrey AL, Mahamdeh M, Tomeh T, Breyne K, Roumieh E, Gurevich M, Soberman RJ, Wojtkiewicz GR, Chen JW, Han JS, Stemmer-Rachamimov A, Breakefield XO
AAV hamartin gene therapy in a stochastic, cerebral mouse model of tuberous sclerosis type 1. Mol Ther Methods Clin Dev. 2025;33(3):101556 - PMID: 40932991 - PMCID: PMC12419009 - DOI: 10.1016/j.omtm.2025.101556Wang C*, Jalali Motlagh N*, Wojtkiewicz GR, Yang H, Kim HH, Chen JW
A specific and adaptable approach to track CD206+ macrophages by molecular MRI and fluorescence imaging. Theranostics. 2025;15(3):1094-1109 - PMID: 39776805 - PMCID: PMC11700851 - DOI: 10.7150/thno.96488Petrozziello T, Motlagh NJ, Monsanto RZB, Lei D, Murcar MG, Penney EB, Bragg DC, Fernandez-Cerado C, Legarda GP, Sy M, Muñoz E, Ang MC, Diesta CCE, Zhang C, Tanzi RE, Qureshi IA, Chen JW, Sadri-Vakili G
Targeting Myeloperoxidase to Reduce Neuroinflammation in X-Linked Dystonia Parkinsonism. CNS Neurosci Ther. 2024;30(11):e70109 - PMID: 39500625 - PMCID: PMC11537767 - DOI: 10.1111/cns.70109Motlagh NJ, Wang C, Kim HH, Jun Y, Kim D, Lee S, Chen JW
Aging Intensifies Myeloperoxidase Activity after Ischemic Stroke. Aging Dis. 2024;15(6):2650-2664 - PMID: 39325942 - PMCID: PMC11567251 - DOI: 10.14336/AD.2023.1640Petrozziello T, Motlagh NJ, Monsanto RZB, Lei D, Murcar MG, Penney EB, Bragg DC, Fernandez-Cerado C, Legarda GP, Sy M, Muñoz E, Ang MC, Diesta CCE, Zhang C, Tanzi RE, Qureshi IA, Chen JW, Sadri-Vakili G
Targeting myeloperoxidase to reduce neuroinflammation in X-linked dystonia parkinsonism. medRxiv. 2024;:ePub - PMID: 38978657 - PMCID: PMC11230314 - DOI: 10.1101/2024.06.25.24309481- More publications ...
Research projects
Imaging Biomarker Discovery for Neuroinflammation
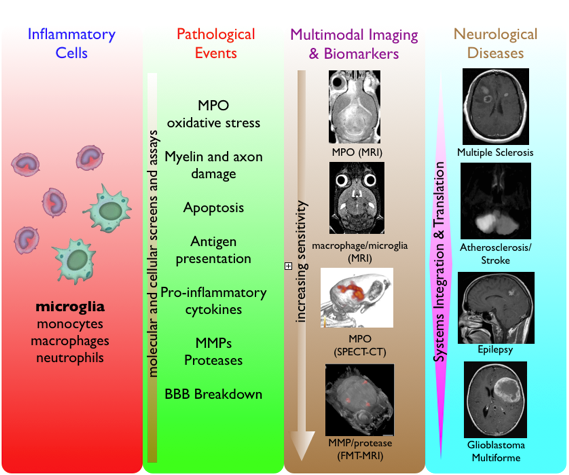
Neuroinflammation occurs with nearly all neurological diseases. The brain is particularly susceptible to inflammation and the resultant oxidative stress as it contains large quantities of oxidizable lipids and metals and has fewer antioxidant mechanisms than other tissues. Important to understanding what roles neuroinflammation has is to be able to track various key components of neuroinflammation in vivo without additional manipulations from ex vivo and in vitro experiments that may temper the biological processes. For example, our research in the development of activatable molecular imaging agents for neuroinflammation has developed multiple agents that can report with high sensitivity and specificity a key enzyme in inflammation, myeloperoxidase (MPO) by both MR and nuclear imaging. MPO is a potent inflammatory mediator found in many activated leukocytes and is postulated to interact with many pro-inflammatory molecules and cells that contribute to neuroinflammation. In vivo applications of these agents have led to improved understanding and the ability to evaluate novel therapies for multiple sclerosis, stroke, atherosclerosis, myocardial infarction, and heart transplant. We seek to develop novel imaging biomarkers as well as integrating existing library of inflammatory imaging sensors to decipher neuroinflammation and its relationship to pathology and disease.
Multiple Sclerosis

Multiple sclerosis (MS) is the leading cause of non-traumatic neurological disability in young adults and MS related health costs are estimated to be more than $10 billion annually in the United States. Despite significant advances in the understanding of MS pathogenesis and high level anatomic imaging by MRI, the initial diagnosis of MS, timely diagnosis of MS relapses, and treatment of MS remain a challenge. Although the etiology of MS remains elusive to this day, there is tremendous evidence that MS is primarily an inflammatory disease and current treatment strategies are based on immune modulation of inflammation. However, there is a lack of an accurate, sensitive, and predictive biomarker to report on active inflammation and demyelination in MS. Currently, MS disease activity is diagnosed using clinical symptoms, invasive CSF analysis, and nonspecific conventional gadolinium chelate enhancement on MR imaging. Using advanced targeted multimodal molecular imaging technology and specific modulations, we study the relationship between key inflammatory enzymes, molecules, and cells to generate a roadmap of potential biomarkers to improve noninvasive diagnosis and to discover opportunities for novel, more effective therapy.
News
CSB investigators - Mikael Pittet, PhD, Matthias Nahrendorf, MD, PhD, and John Chen, MD, PhD - have been named Distinguished Investigators of the Academy of Radiology Research. Congratulations!
Benjamin Pulli, MD from the Chen Lab has been chosen by the RSNA Scientific Program Committee to receive the RSNA Travel Award for Young Investigators in Molecular Imaging for abstract "Molecular Magnetic Resonance Immunoradiology Reveals Novel Effect of Interferon-? on Myeloid Cells in Murine Multiple Sclerosis".
"Imaging Early MS" - An Interview with John Chen, M.D., Ph.D. by Dana Foundation (pdf)
Benjamin Pulli's work on distinguishing inflammatory liver disease from benign fatty liver was featured in the RSNA Daily Bulletin. (pdf)
Congressionally Directed Medical Research Programs site highlights work of John Chen, M.D, Ph.D. - New Imaging Agent for Early Lesion Detection and Treatment Tracking in MS. (pdf)