Lin Lab
Read BioThe main research focus of our group is to develop minimally invasive optical techniques for in vivo imaging and monitoring of cells and tissues as well as therapeutic applications of lasers. The diagnostic techniques will help to answer important biological questions, such as:
- In-vivo monitoring of cell trafficking in circulation
- Imaging of vasculature and microenvironment in tissue
- Interaction of cells with microenvironment
The therapeutic techniques allow us to target cellular and subcellular structures by means of selective absorption of endogenous or exogenous chromophores. Selective targeting may be helpful for treating various pathologies, such as retinal diseases or destruction of tumors, without causing adverse side effects to healthy tissue.
Recent Publications
Gustafsson K, Isaev S, Mirsanaye K, Hofmann J, Kooshesh KA, Baryawno N, Kiem A, Severe N, Zhao T, Scadden EW, Spencer JA, Burns C, Akilan K, Barkas N, Gessessew H, Kokkaliaris KD, Lin CP, Kharchenko PV, Scadden DT
Mesenchymal thymic niche cells enable regeneration of the adult thymus and T cell immunity. Nat Biotechnol. 2025;:ePub - PMID: 41168496 - DOI: 10.1038/s41587-025-02864-wPessoa Rodrigues C, Collins JM, Yang S, Martinez C, Kim JW, Lama C, Nam AS, Alt C, Lin C, Zon LI
Transcripts of repetitive DNA elements signal to block phagocytosis of hematopoietic stem cells. Science. 2024;385(6714):eadn1629 - PMID: 39264994 - PMCID: PMC12012832 - DOI: 10.1126/science.adn1629Wu Q, Zhang J, Kumar S, Shen S, Kincaid M, Johnson CB, Zhang YS, Turcotte R, Alt C, Ito K, Homan S, Sherman BE, Shao TY, Slaughter A, Weinhaus B, Song B, Filippi MD, Grimes HL, Lin CP, Ito K, Way SS, Kofron JM, Lucas D
Resilient anatomy and local plasticity of naive and stress haematopoiesis. Nature. 2024;627(8005):839-846 - PMID: 38509363 - PMCID: PMC10972750 - DOI: 10.1038/s41586-024-07186-6Conedera FM, Kokona D, Zinkernagel MS, Stein JV, Lin CP, Alt C, Enzmann V
Macrophages coordinate immune response to laser-induced injury via extracellular traps. J Neuroinflammation. 2024;21(1):68 - PMID: 38500151 - PMCID: PMC10949579 - DOI: 10.1186/s12974-024-03064-0Bonora M, Morganti C, van Gastel N, Ito K, Calura E, Zanolla I, Ferroni L, Zhang Y, Jung Y, Sales G, Martini P, Nakamura T, Lasorsa FM, Finkel T, Lin CP, Zavan B, Pinton P, Georgakoudi I, Romualdi C, Scadden DT, Ito K
A mitochondrial NADPH-cholesterol axis regulates extracellular vesicle biogenesis to support hematopoietic stem cell fate. Cell Stem Cell. 2024;31(3):359-377.e10 - PMID: 38458178 - PMCID: PMC10957094 - DOI: 10.1016/j.stem.2024.02.004- More publications ...
Research projects
TECHNOLOGY
Multimodal Microscopy
Imaging modalities such as confocal reflectance microscopy, fluorescence by single- or two-photon excitation, second harmonic generation and coherent anti-Stokes Raman spectroscopy (CARS) provide different contrast mechanisms that can be combined to give structural, functional and molecular information of living tissue. Our group has developed a multimodal microscope in which up to three of these imaging modalities can be realized simultaneously. Images are acquired at video rate, which allows real-time monitoring of fast events in the living tissue.
Adaptive Optics
Ophthalmic imaging resolution suffers from aberrations present in the eye. With the introduction of an adaptive optics (AO) system into a scanning laser ophthalmascope (SLO) one can achieve the best possible resolution for the system. We have implemented such an AO system into a SLO designed for imaging the mouse retina.
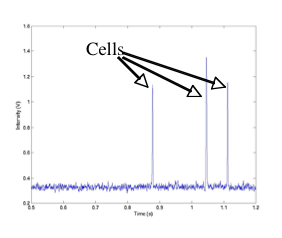
In vivo flow cytometry
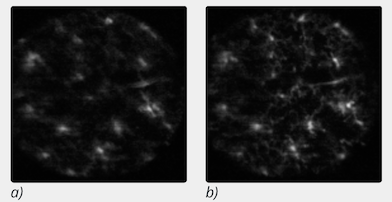
(a) Aberrated image (AO system off).
(b) Corrected image (AO system running)
BIOLOGICAL STUDIES
Vascular Biology
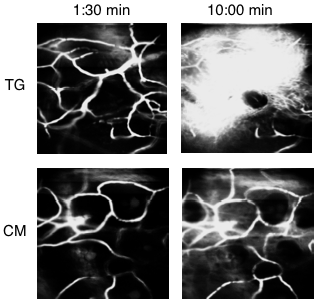
Role of IKK-ß in Akt activation and vascular permeability. Evans blue dye was injected into a transgenic (TG) and control mouse (CM). Images were taken at 1:30 and 10:00 minutes after injection. Increased vessel leakage was shown in the TG mouse, due to the deletion of IKK-ß, which reduces activation of Akt, and increases vascular permeability.
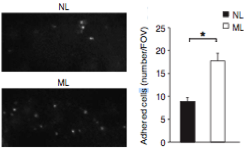
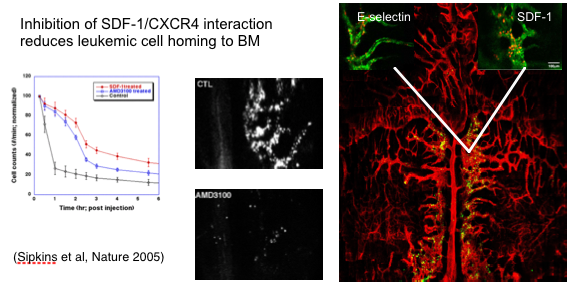
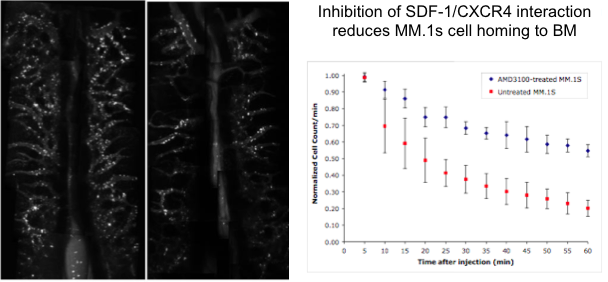
Cancer
Transplantation Immunology
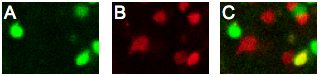
A) Image of regulatory T cells (FOXP3-GFP) that have migrated to the islet graft after adoptive transfer. B) Image of effector T cells (U-DsRed) acquired at the same time as A. C) Co-registered image revealing natural regulatory T cells (green), effector T cells (red), and a converted regulatory T cell (yellow).
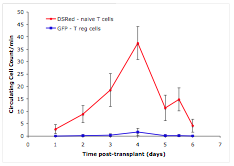 Simultaneous, independent detection of GFP+ regulatory T cells (blue) and DSRed+ naïve T cells (red).
Simultaneous, independent detection of GFP+ regulatory T cells (blue) and DSRed+ naïve T cells (red).
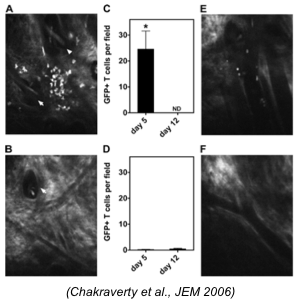
Investigation of T cell migration in graft versus host disease (GVHD). Images of transplanted T cells near a hair follicle in a (A) freshly irradiated mouse and (B) mixed Chimera (MC) mouse 5 days after transplantation. T cell counts after 5 and 12 days in the irradiated mouse (C) and MC (D). Images of T cells 5 days after transplantation near postcapillary venules in the freshly irradiated mouse (E) and the MC (F).
Stem Cell Biology
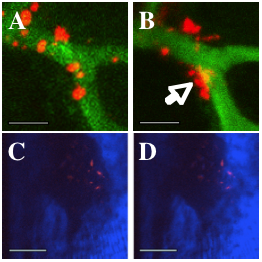 In vivo confocal and 2-photon microscopy demonstrating the extravasation of FTVI treated MSCs and subsequent migration to the bone surface in the mouse bone marrow after adoptive transfer. DiD-labeled MSCs (red), primarily line the vessel walls (green) at 1 hr after transfer A), whereas at 24 hrs B), the MSCs show a marked increase in extravasation (arrow). Second Harmonic Generation microscopy of bone (collagen, blue) reveals the close juxtaposition between the cells and the bone after 9 days. The bone image in D) is 10µm above the image in C). Bar=50µm for A-B) and 100µm for C-D).
In vivo confocal and 2-photon microscopy demonstrating the extravasation of FTVI treated MSCs and subsequent migration to the bone surface in the mouse bone marrow after adoptive transfer. DiD-labeled MSCs (red), primarily line the vessel walls (green) at 1 hr after transfer A), whereas at 24 hrs B), the MSCs show a marked increase in extravasation (arrow). Second Harmonic Generation microscopy of bone (collagen, blue) reveals the close juxtaposition between the cells and the bone after 9 days. The bone image in D) is 10µm above the image in C). Bar=50µm for A-B) and 100µm for C-D).
News
Three CSB researchers - Clemens Alt, PhD (Lin Lab), Cameron McAlpine, PhD and Shun He, PhD (Swirski Lab) - won Poster of Distinction award at the Annual Meeting of the MGH Scientific Advisory Committee (SAC). Congratulations!
Clemens Alt, PhD won the 2012 Pascal Rol Award for Best Paper in Ophthalmic Technologies at the recent SPIE Photonics West, BiOS conference in San Francisco for his presentation and proceedings paper entitled "In vivo quantification of microglia dynamics with a scanning laser ophthalmoscope in a mouse model of focal laser injury". (pdf)
"Color-coded Mouse Sheds Light on Transplant Tolerance" - work of Charles Lin's Lab is featured in the Harvard Focus. (pdf)